Experimental drug offers new hope for coeliac disease treatment – protects the whole body from the harmful effects of gluten

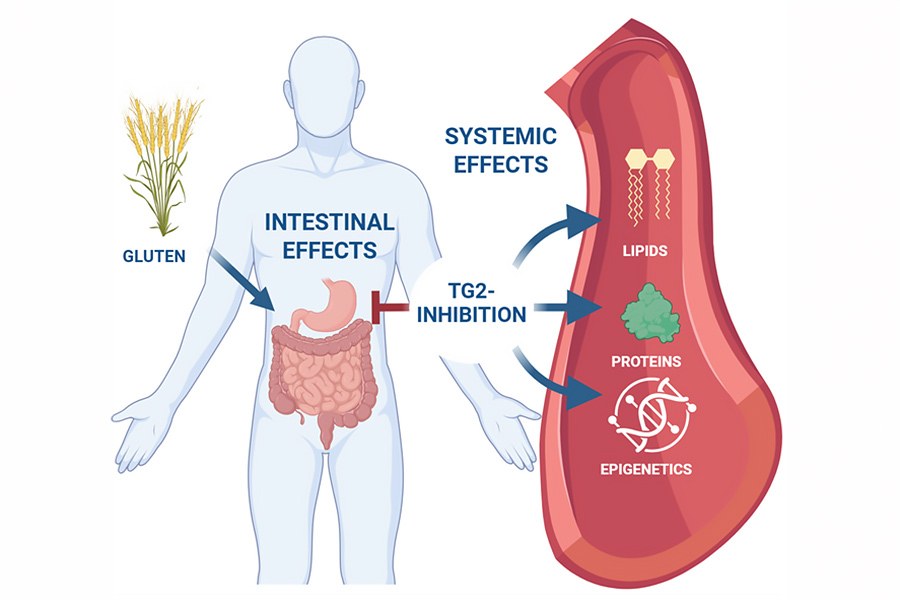
The drug examined in the study, ZED1227, inhibits the activity of the body’s transglutaminase 2 (TG2) enzyme. This enzyme modifies gluten in a way that triggers an inflammatory response in coeliac disease. By blocking the enzyme, the drug reduces the harmful immune responses caused by gluten.
The new study was published in the BMC Medicine journal and expands on earlier findings. In a study published in Nature Immunology in 2024, the same research group showed that a TG2 inhibitor effectively protects the small intestinal mucosa from gluten-induced damage. The latest research examines, for the first time, its effects at the level of the whole body.
The study involved adults with coeliac disease who had long adhered to a gluten-free diet. They were exposed to small amounts of gluten for six weeks, either in combination with the drug or with a placebo.
Blood samples were analysed for lipid metabolism, proteins and DNA methylation. These were compared with tissue samples taken from the small intestine. The results showed that gluten caused clear changes in lipid metabolism in the placebo group, whereas in those receiving the drug, the changes were almost completely suppressed. The experimental drug also restored blood protein and epigenetic profiles to resemble those seen during a gluten‑free diet.
The findings suggest that TG2 inhibition has effects beyond the gut and dampens the whole-body immunometabolic responses triggered by gluten. This is a new and scientifically significant discovery.
“Our study published in 2024 showed that the drug effectively protects the intestinal mucosa. We now demonstrated that its effects extend to the whole body,” says the corresponding author of the study, Associate Professor Keijo Viiri.
According to the researchers, the drug could in future complement dietary treatment, particularly in patients who are inadvertently exposed to gluten or whose symptoms persist despite a gluten-free diet.
Viiri’s research group is currently investigating how inflammation-induced epithelial damage could be prevented or repaired through drug therapy in people with coeliac disease. The aim is to strengthen the protective effect of treatment, especially in high-risk patients.
Coeliac disease is an autoimmune condition in which gluten triggers an inflammatory response in genetically susceptible individuals. At present, the only treatment is a lifelong gluten-free diet, which can be challenging to maintain. In Finland, around 2.4 per cent of the population has coeliac disease, one of the highest prevalence rates in the world.
The study is part of an international clinical trial and has been funded, among others, by the pharmaceutical company Dr. Falk Pharma GmbH. It is also part of the University of Oulu’s Profi8 Health Dimensions research programme, funded by the Research Council of Finland.
The study Therapeutic TG2 inhibition reverses systemic multiomic dysregulation in celiac disease was published in the BMC Medicine journal on 24 April 2026. Read the article
Read the group’s previous article in Nature Immunology: Transcriptomic analysis of intestine following administration of a transglutaminase 2 inhibitor to prevent gluten-induced intestinal damage in celiac disease.





