Researchers create a new human derived adipose tissue model that helps to understand obesity without animal testing

Obesity and associated diseases – including cardiovascular diseases, type 2 diabetes, and several cancers – cause millions of deaths annually. Much research is being conducted on adipose tissue and obesity, but the challenge lies in the way the research is carried out. Nearly all adipose tissue and obesity studies are conducted using animal models that poorly correspond to the human body and its functions.
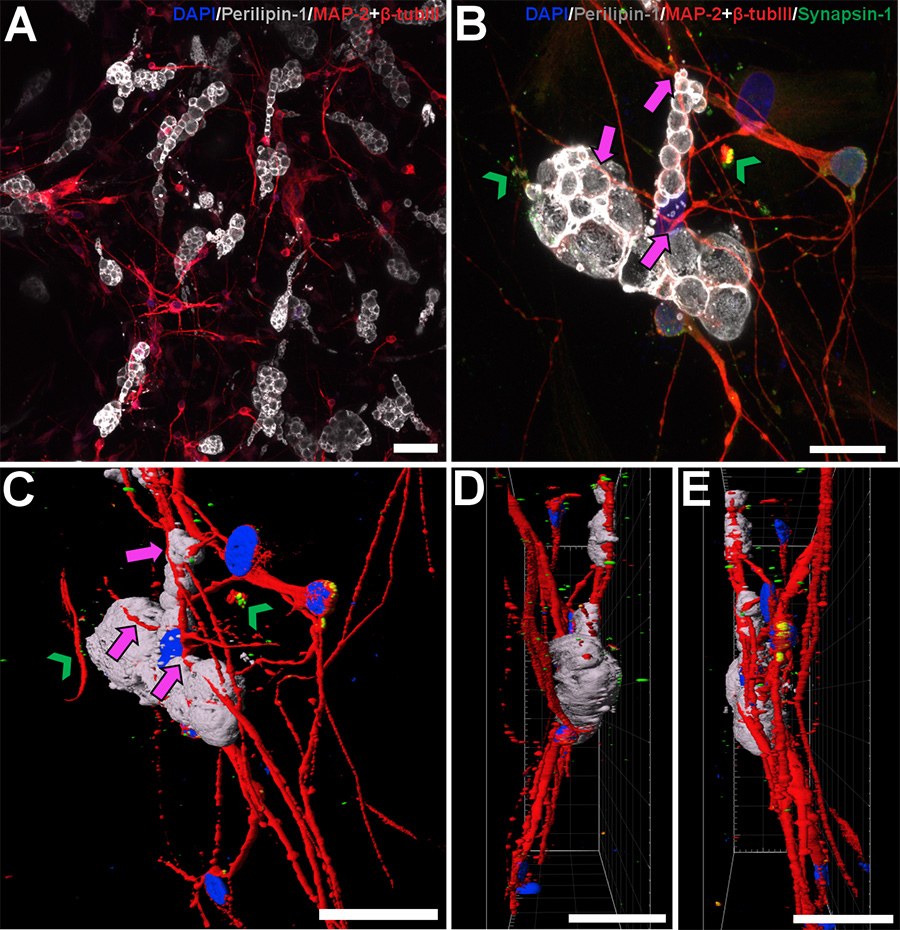
In the future, however, research may be conducted in a better way. Doctoral researcher Sini Saarimaa and the research group where she works have developed a three-dimensional human derived in vitro adipose tissue model that can be used in obesity research.
Saarimaa focuses particularly on developing and studying the connection between the nervous system and adipose tissue. For many, it may come as a surprise that the central nervous system, meaning the brain and spinal cord, is known to regulate the function of adipose tissue together with the peripheral nervous system. Does this suggest that weight could be regulated purely by mental effort?
“Weight management is determined by a complex interaction between biology, behaviour, and the environment, meaning that just thinking about weight less is not sufficient,” Saarimaa explains.
Adipose tissue is active and essential for the body’s energy balance and metabolism. It requires both neural and hormonal signalling to function normally. For example, functional disorders in the nervous system have been linked to the development of overweight and obesity.
The tissue model developed by Saarimaa et.al. provides new information on the interactions between the nervous system and adipose tissue. In particular, it enhances understanding the role of the central nervous system in promoting adipose stem cell maturation, metabolic activity and functional signalling.
Adipocytes are active communicators
What astonished Saarimaa the most was discovering how active the maturing cells in adipose tissue – adipocytes – are. One of the methods used in the study was imaging the electrical activity of neurons by observing the calcium spikes they produce, which appear under the microscope as blinking fluorescent signals inside the cells.
“We noticed that adipocytes also exhibit ‘blinking’, meaning they too presented active calcium-mediated signalling. That was a very positive surprise,” Saarimaa recalls.
The surprise was all the greater because research rarely offers sudden, television style aha moments and results typically arise from long-term, tenacious work. In this project as well, the findings were validated using cell lines isolated from different patients, which meant repeating the assays numerous times for each analysis.
In upcoming research, the model will be further developed by examining the density and functionality of innervation. This will include studying, for example, the transmission of electrical nerve impulses to nerve endings and the effects of neurotransmitters released from the nerve endings on adipocytes.
“We will add human peripheral nervous system neurons and mature adipocytes, bringing our adipose tissue model even closer to the structure of human adipose tissue,” Saarimaa explains.
The developing tissue model may help to identify new drug molecules that act directly on adipose tissue. It can also help reduce the use of animal models, as it provides a more relevant, human-based research platform that supports the development of better prevention and treatment methods. At the same time, it advances personalised medicine in the treatment of obesity and related metabolic diseases.
The article Human induced pluripotent stem cell-derived neurons and coculture conditions regulate the adipogenic differentiation and functionality of human adipose stromal/stem cells was published in the Cell Communication and Signaling journal in November 2025. The article was authored by Sini Saarimaa, Miia Juntunen, Lotta Isosaari, Reija Autio, Marika Kuuskeri, Susanna Narkilahti, Susanna Miettinen. The article is available online
The main funders of the research include the Research Council of Finland, Tampere University Hospital/wellbeing services county of Pirkanmaa, Finnish Cultural Foundation, Finnish Diabetes Association and Tampere University’s Institute for Advanced Study.





