Cleanroom facility

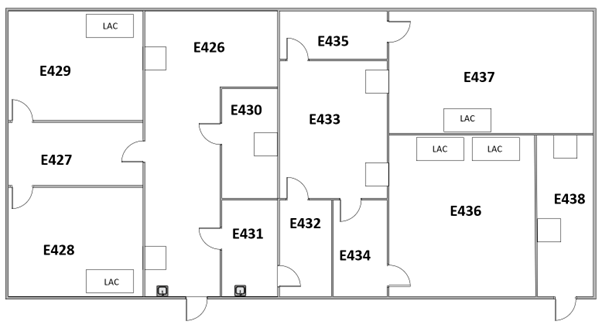
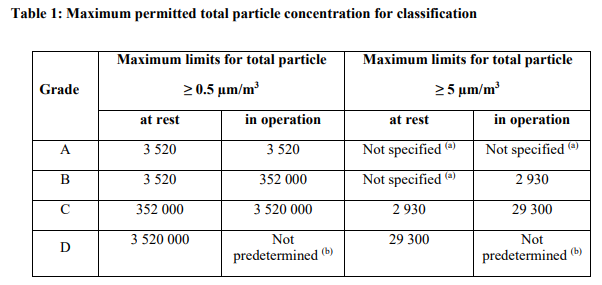
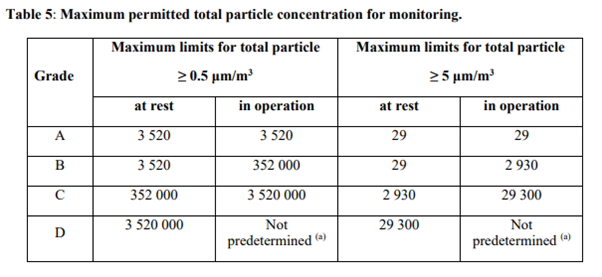
MET cleanroom facility is located on Arvo building 4th floor. It has two production suites (GMP A/B classification) and two production suites (GMP A/C classification) with adjacent QC laboratories specifically designed for the processing and manufacture of tissue and cell-based products. Laminar cabinets are available in the cleanroom suites. A quality control / quality assurance program with cleanroom and equipment qualifications, an online facility monitoring system as well as cleaning and environmental monitoring procedures is in place. There is also a possibility for materials storage (quarantine / released).
The cleanroom suites may be rented out to internal or external users. Contract manufacture and service activities are also available. Training will be provided to all cleanroom users.
Regea Tissue Center at MET is responsible for coordinating the cleanroom activities. Regea uses part of the cleanroom for its own tissue processing activities. Regea holds a tissue establishment license and is inspected regularly by Fimea (Finnish Medicines Agency).
More information on Regea: