Cells respond quickly to small light-induced micro-environment movements

Three research groups investigated in their joint project how epithelial cells sense small changes in their environment through ion channels. The study was conducted using light-responsive materials developed by the Smart Photonics Materials research group led by Professor Arri Priimägi, which can be used as a substrate for cell culturing. These materials allow precise and controllable movement of the cell substrate using light stimulation.
“The cells had a marker protein for intracellular calcium, so we were able to draw small grooves on the substrate surface on a confocal microscope and at the same time monitor how living cells respond to these changes in the environment with the help of calcium. We found that even the movement of a few tens of nanometres of material opened mechanically gated calcium channels in the cells, through which the cells were able to change their calcium levels,” says Teemu Ihalainen, Senior Research Fellow at Tampere Institute for Advanced Study (IAS) and leader of the Cellular Biophysics research group at the Faculty of Medicine and Health Technology.
Calcium is needed in cells for a wide variety of processes, so even small changes in the amount of calcium can have large effects on cellular functions. The study shows, perhaps for the first time, that cells are able to sense minute movements in their environment and these movements are detected by changing the flow of calcium ions through the cell membrane, i.e. electrically via ionic currents.
The study focused on intracellular calcium changes during the first seconds of the mechanical stimulus. An article titled Light-induced nanoscale deformation in azobenzene thin film triggers rapid intracellular Ca2+ increase via mechanosensitive cation channels , which is a key part of Doctoral Researcher Heidi Peussa’s dissertation, was published in November 2023 in the prestigious journal Advanced Science.
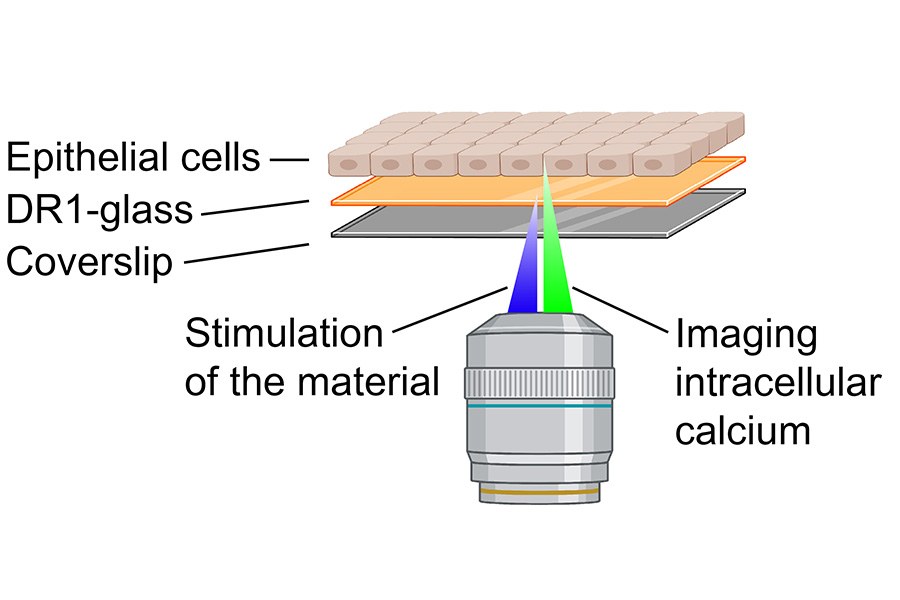
Mechanically gated ion channel as a key
In the body, epithelial cells are tightly attached to extracellular matrix, allowing mechanical strain of the environment, for example, to be transmitted to the cells. Mechanical stimuli are important in the normal functions of cells. Disruption of cell attachment often causes disease or other problems.
Cells sense changes in their environment in a variety of ways, for example, by mechanically gated PIEZO1 ion channels. The channels can be understood as cell membrane pores that are closed in mechanically relaxed state, but open as the cell membrane stretches. The opening happens in thousandths of a second and leads into calcium influx to the cell. The process has a key role in many physiological functions, e.g., in touch sensation. The discovery of mechanically gated ion channels was awarded the Nobel Prize in 2021.
The study showed that PIEZO1 channels are crucial for sensing rapid changes in the cell’s microenvironment.
“We discovered that cells are capable of sensing deformations as small as 40 nanometres (0.000040 mm) that occur in thousandths of a second. For the first time, we were able to monitor how the PIEZO1 channel opens as a result of a physical change in the local, extracellular environment,” Ihalainen mentions.
New possibilities to study cellular processes of the eye
The method used by the researchers is new and allows studying the mechanical stimulus of the extracellular matrix in particular and simultaneously monitoring cell responses. Further research addressing the PIEZO1 channel functioning are already on the way. In addition, the researchers aim is to study and develop new light-responsive materials.
“Our next steps are to study the regulation and regulating factors of these mechanically gated ion channels. The goal is also to gain a broader understanding of what happens after the first few seconds in the perception of force sensation. We are developing new transgenic cell lines to further study calcium signalling at different locations in the cell. These transgenic cell lines also allow us to extend the studies to underlying retinal pigment epithelium of the eye and to study the role of PIEZO1 channels in retinal maintenance,” says Soile Nymark, Associate Professor of Biosensor technology and leader of the Biophysics of the Eye research group at the Faculty of Medicine and Health Technology.
The study published is part of the three-year ABioT project funded by the Emil Aaltonen Foundation. The project has also received funding from the Research Council of Finland and Tampere Institute for Advanced Study (IAS).
Further information
Teemu Ihalainen
+358 50 318 7202
teemu.ihalainen [at] tuni.fi
Heidi Peussa
heidi.peussa [at] tuni.fi
Soile Nymark
soile.nymark [at] tuni.fi
Arri Priimägi
arri.priimagi [at] tuni.fi
Author: Anna Aatinen





